
Case study
Optical diagnostic device for intracranial haemorrhage detection
Systems-led technical leadership converting a performance-sensitive concept into a robust, ICU-ready medical device.
Engagement summary
The challenge
Stabilising a noise-sensitive optical concept so it performs reliably in real ICU conditions, with usability and safety constraints of a Class IIb pathway.
Approach
Whole-system optimisation across optics, mechanics, electronics, software, and workflow, with risk controls and rapid verification cycles.
Outcomes
Fivefold signal-to-noise improvement, coupling time cut from ~5 minutes to <1 minute, and an ICU-deployed pilot with defensible technical evidence.

“Working with Earle gave us confidence the system integrity was in safe hands. He kept focus on clinical reality and patient impact, guiding rapid design and test iterations with sound technical judgment. That balance of rigour and pragmatism substantially improved reliability and usability as we moved into clinical evaluation.”
Barry Dixon, ICU clinician and Founder, Wavewise Analytics
The challenge: translating technical promise into stable clinical performance

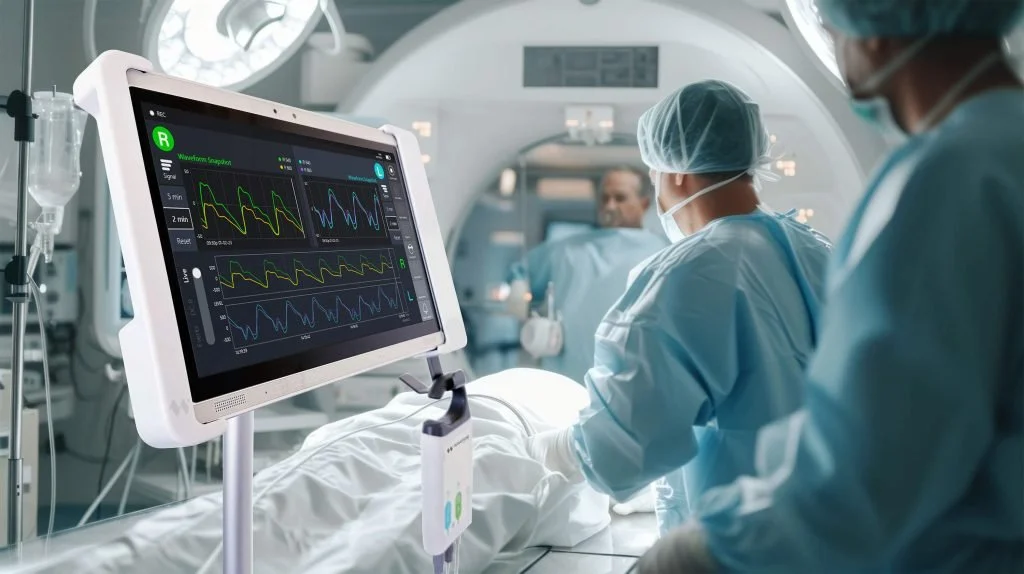
Wavewise Analytics is a MedTech innovator focused on continuous, non-invasive brain monitoring for early detection of neurological deterioration in critical care.
Translating this optical innovation into a reliable bedside system revealed significant sensitivity to noise, coupling stability, mechanical tolerances, and biological variability. Stabilising system performance was essential before clinical deployment.
Founded by a critical care surgeon and supported by a multidisciplinary team, the objective was to translate the concept into a coherent, safe, and usable clinical system within six months, aligned with a Class IIb diagnostic pathway.
Key risks included:
Unstable signal acquisition under real-world conditions
Usability constraints within high-pressure ICU workflows
Early architectural decisions that could constrain future regulated development
Addressing these risks required disciplined systems leadership to stabilise system behaviour, integrate cross-disciplinary design decisions, and establish a defensible technical foundation for clinical evaluation.
Approach: whole-system optimisation under clinical and regulatory constraints
A disciplined systems approach enabled rapid iteration while maintaining control over safety, usability, and performance.
Use specification as anchor
Developed an IEC 62366-1–aligned use specification defining intended users, ICU conditions, workflow integration, and performance boundaries, preventing early design misalignment.
Whole-system performance focus
Mapped the full measurement chain from optical emission to algorithm output to identify dominant sources of variability across mechanical, optical, and electronic domains. Delivered 10+ transducer housing iterations verified on controlled reference surfaces. Results included a fivefold improvement in signal-to-noise ratio and reduced coupling time from ~5 minutes to under 1 minute, with improved robustness across scalp and positioning variability.
Risk and essential performance
Led ISO 14971–aligned hazard analysis and defined essential performance characteristics, implementing controls including signal adequacy detection, positioning prompts, and IEC 60601-1 / -1-2 electrical safety and EMC alignment. Development decisions assumed a regulated diagnostic pathway, avoiding future technical debt.
Multidisciplinary integration
Coordinated mechanical, electrical, and software disciplines through continuous integration and verification cycles, ensuring system behaviour remained the optimisation target.
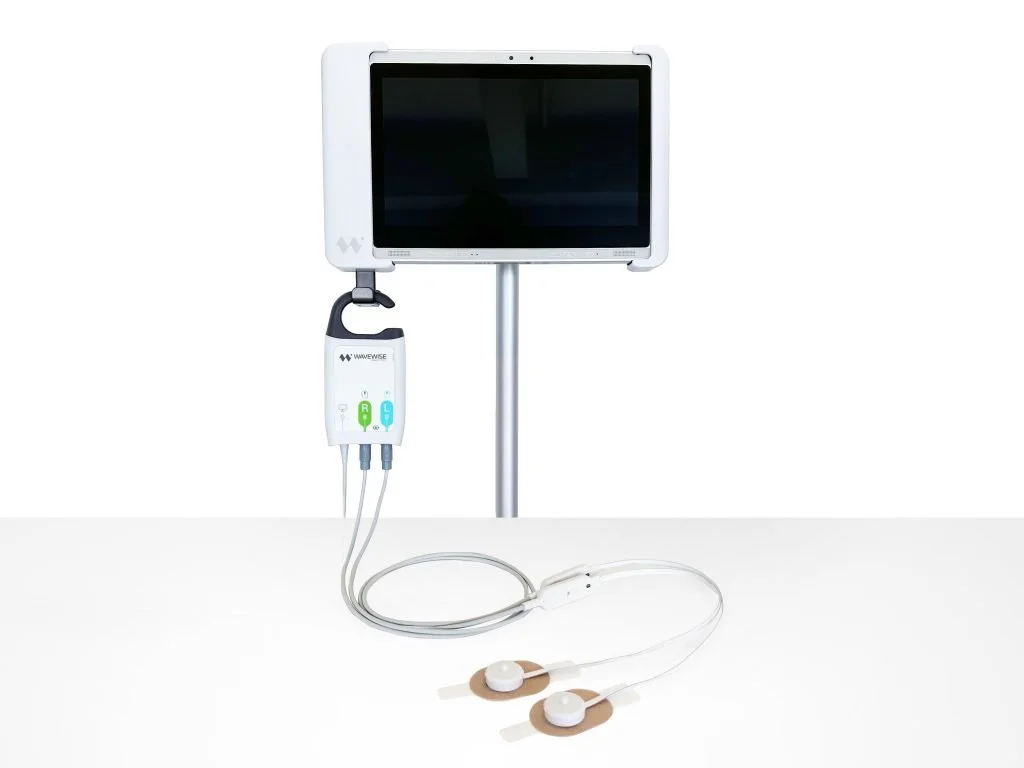
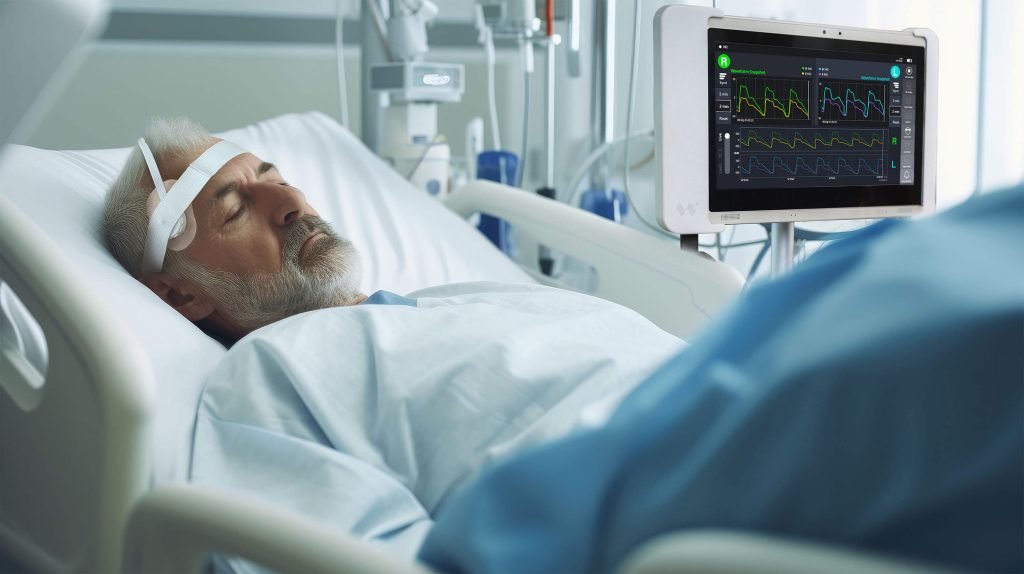
Outcome: a stable clinical pilot platform with demonstrable performance gains
The programme delivered an optical monitoring system deployed in an ICU for structured pilot evaluation under clinical supervision.
Key outcomes:
Translated a noise-sensitive research concept into a stable and repeatable clinical measurement platform
Demonstrated quantified performance improvement through structured, multidisciplinary iteration
Reduced setup variability and improved operational reliability within a real ICU workflow
Defined essential performance characteristics linking system behaviour directly to clinical safety
Established a scalable product architecture aligned with clinical and regulatory requirements.
This engagement shows how structured systems leadership converts technical feasibility into safe, usable clinical performance, grounding engineering decisions in clinical context and regulatory constraints while maintaining rapid iteration.
