
Engineering a Focused Ultrasound Platform for Neurological MedTech Research
Case study
Translating world-class neuroscience research into a controlled platform for targeted ultrasound delivery.
Summary
The Queensland Brain Institute Clem Jones Centre for Ageing Dementia Research is home to world-class research into ultrasound-based therapies for neurological disease. Led by Prof Jürgen Götz, the team had established therapeutic potential through research prototypes. The next challenge was moving from sonication capability toward a controlled pre-clinical research platform capable of targeting specific brain regions, controlling pressure delivery, accounting for patient-specific variability, and supporting future medical technology development.
Client: Queensland Brain Institute, University of Queensland
Sector: Medical Technologies, Neurological Research
Product type: MedTech/Research Platform
Stage: Translational research (pre-clinical)
Role: Engineering leadership, systems engineering, technical coordination
Challenge: Translating prototypes into a controlled, patient-specific platform for targeted pre-clinical research
Outcome: A pre-clinical research platform for targeting and dose delivery, with a preliminary design and safety case to support future medical technology development.

“Earle understood the science and what we needed from an engineering perspective. He brought medical technology development experience, structure, and leadership to a complex challenge, leading the engineering team to develop our next-generation technology. This was instrumental in advancing our research and clarifying the path toward a future medical device.”
Jürgen Götz, Professor, Director, Clem Jones Centre for Ageing Dementia Research, QBI, University of Queensland
The Engineering Challenge
QBI had established promising focused ultrasound research for neurological disease, supported by early prototype capability.
The challenge was to develop a platform that could support more controlled, repeatable, and targeted ultrasound delivery for ongoing pre-clinical investigation.
This meant moving beyond proof-of-concept sonication toward an integrated system that could connect targeting, pressure delivery, positioning, clinical workflow, and safety in a way that supported both research use and future medical technology development.
Conceptual illustration of the QBI focused ultrasound delivery system
Approach
This engagement used a systems engineering approach to translate QBI’s research objectives into a pre-clinical platform for ongoing focused ultrasound research.
The work began by defining the use specification, user needs, design inputs, and system architecture, then coordinating parallel engineering streams through detailed design, integration, verification, and validation.
An Agile delivery approach was used, using successive prototypes to test assumptions, improve the system, and respond to emerging technical and research insights.
Lightweight ISO 13485 design controls were applied to support traceability from user needs through design, verification, and validation, helping establish an early safety and efficacy case and a clear pathway for future medical technology development.
The result was a controlled research platform that enabled QBI’s ongoing pre-clinical investigation, while also establishing preliminary design and safety-case foundations for future product development.
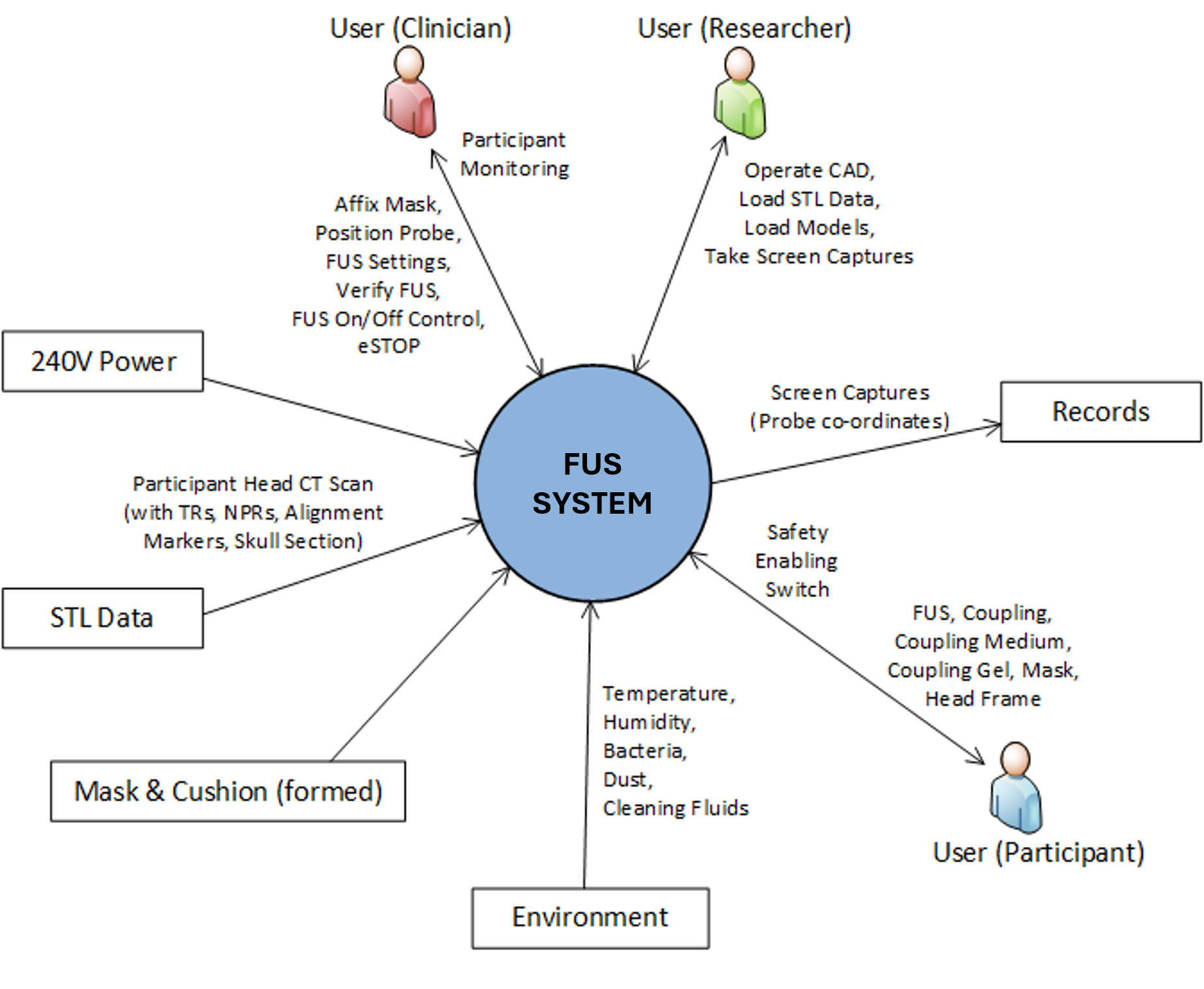
System context diagram. Maps users, inputs, outputs, and interfaces to define the system boundary and operating context.
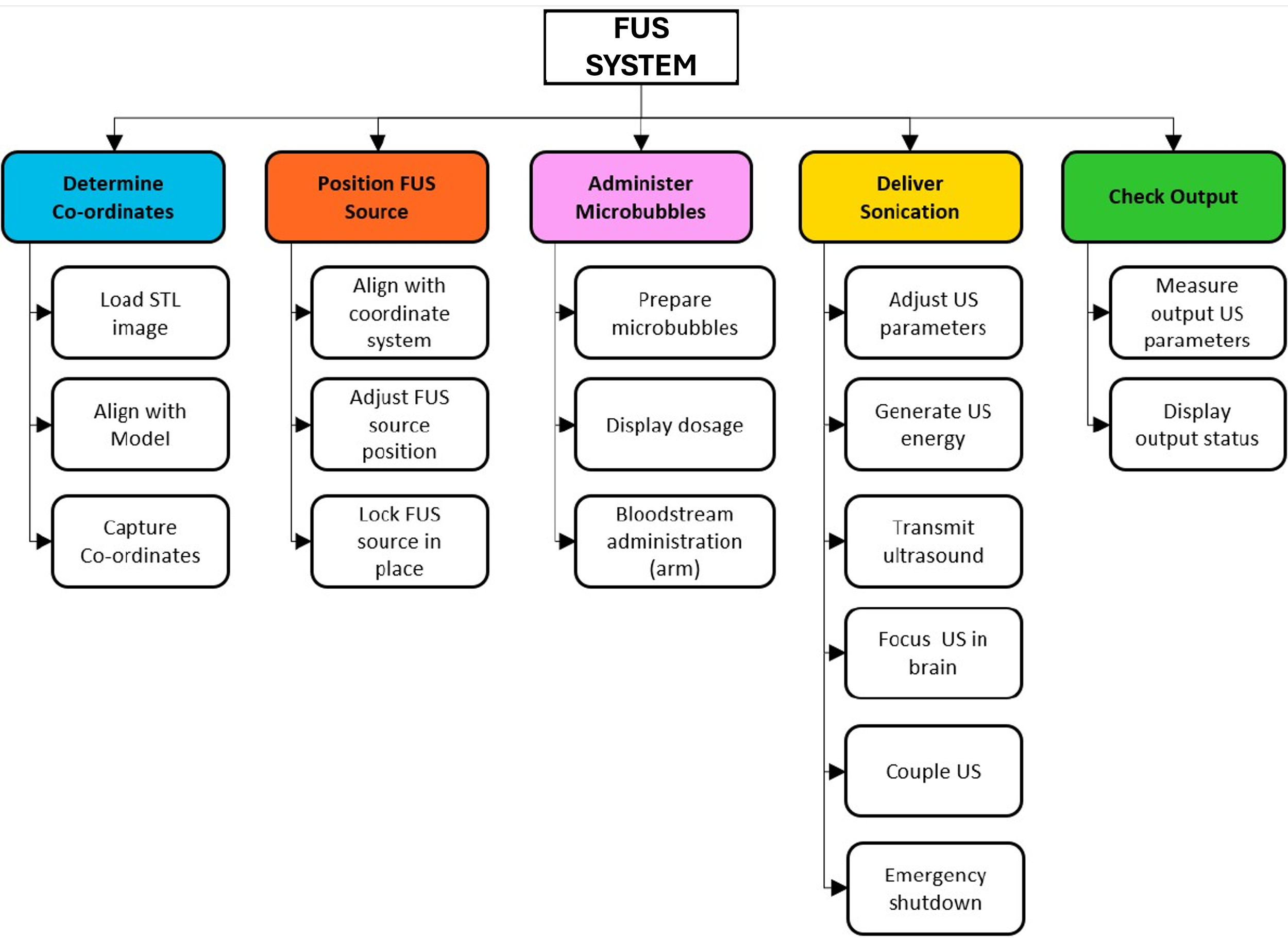
Functional breakdown. Defines the key system functions used to inform requirements, architecture, and development streams.
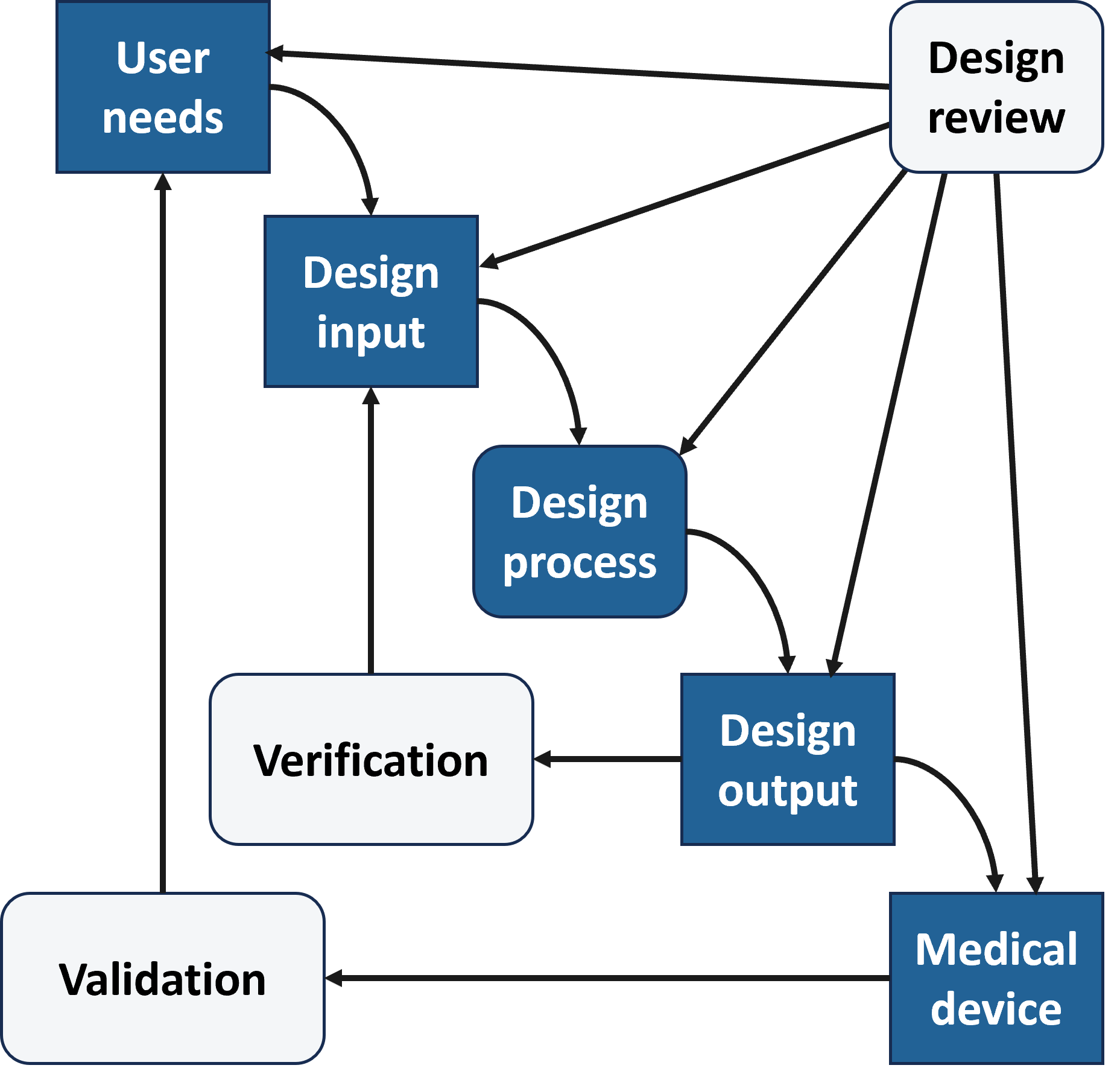
ISO 13485 Design controls tracing user needs through to validation (tool used: Jama)
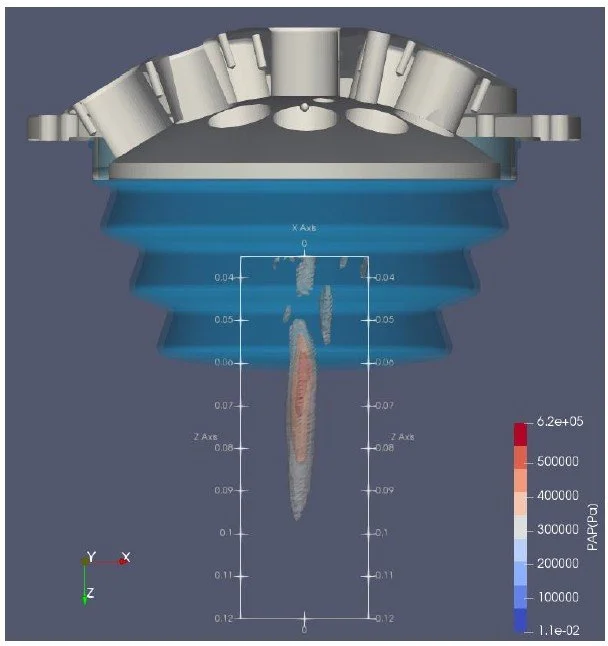
Computational ultrasound model. Calculates sonication pressure distribution to support targeting, pressure delivery, and safety analysis.
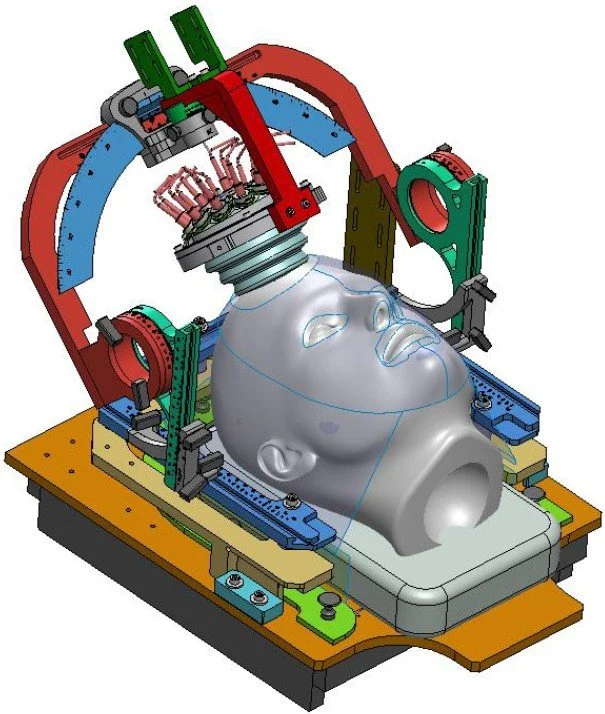
CAD model of the ultrasound positioning and delivery system.
Outcome
This engagement delivered a next-generation pre-clinical research platform that enabled QBI to continue advancing its focused ultrasound research with greater control, repeatability, and confidence.
The platform supported accurate focusing and steering of ultrasound energy, improved confidence in pressure delivery, and provided a more structured basis for experimental investigation.
It also established preliminary design and safety-case foundations for future medical technology development, supported by lightweight ISO 13485 design controls and an Agile prototyping approach that progressively tested and improved the system.

Delivered ultrasound positioning and delivery system.
This engagement illustrates the value of an integrative systems approach in complex MedTech development, bringing together science, regulatory strategy, clinical need, human factors, and engineering to translate ambitious research into an integrated system.
